Japan Pharmaceutical Manufacturing Localization Market to Reach $21.33B by 2033, Driven by Supply Chain Resilience
Government incentives, biologics demand, and domestic API production accelerate Japan’s shift toward localized pharma manufacturing.
Japan’s focus on self-reliant drug production and advanced biologics is reshaping its pharmaceutical ecosystem for long-term resilience and innovation.”
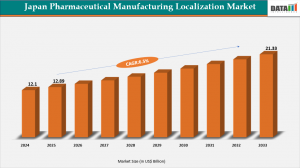
AUSTIN, TX, UNITED STATES, February 23, 2026 /EINPresswire.com/ -- Japan pharmaceutical manufacturing localization market represents a strategic shift towards domestic production of drugs and active pharmaceutical ingredients (APIs), reducing reliance on imports amid global supply chain vulnerabilities. According to DataM Intelligence, the market reached US$12.1 billion in 2024, grew to US$12.89 billion in 2025, and is projected to hit US$21.33 billion by 2033 at a CAGR of 6.5%.— DataM Intelligence
𝗚𝗲𝘁 𝗮 𝗦𝗮𝗺𝗽𝗹𝗲 𝗣𝗗𝗙 𝗕𝗿𝗼𝗰𝗵𝘂𝗿𝗲 𝗼𝗳 𝘁𝗵𝗲 𝗥𝗲𝗽𝗼𝗿𝘁 (𝗨𝘀𝗲 𝗖𝗼𝗿𝗽𝗼𝗿𝗮𝘁𝗲 𝗘𝗺𝗮𝗶𝗹 𝗜𝗗 𝗳𝗼𝗿 𝗮 𝗤𝘂𝗶𝗰𝗸 𝗥𝗲𝘀𝗽𝗼𝗻𝘀𝗲):
https://www.datamintelligence.com/download-sample/japan-pharmaceutical-manufacturing-localization-market
Key growth drivers include post-COVID supply chain resilience efforts, government incentives for onshoring production, surging demand for biologics and advanced therapies, and Japan's rigorous PMDA regulatory standards that favor local quality control. Small-molecule drugs lead the product segments with 40% share due to mature infrastructure and steady demand, while biologics at 30% grow fastest thanks to monoclonal antibodies and biosimilars. The Kanto region, home to Tokyo's biotech clusters and major firms like Takeda, dominates geographically, supported by superior infrastructure, R&D hubs, and policy support.
Key Highlights from the Report
➤ Japan pharma manufacturing localization market to reach US$21.33 billion by 2033 at 6.5% CAGR.
➤ Small-molecule drugs hold 40% share; biologics expanding rapidly at 30%.
➤ In-house manufacturing leads at 45%, followed by CMOs at 35%.
➤ Oncology therapeutics dominate with 30% share amid rising cancer cases.
➤ Government policies boost domestic APIs and supply chain security.
➤ Takeda and Astellas drive investments in biologics and smart factories.
Market Segmentation
By product category, the Japan pharmaceutical manufacturing localization market divides into small-molecule drugs (40% share), biologics (30%), vaccines (15%), cell and gene therapies (10%), and others (5%), with small-molecules leading due to established infrastructure and consistent demand for generics and essentials, while biologics surge via local monoclonal antibody and biosimilar production.
By manufacturing model, segments include in-house manufacturing (45%), contract manufacturing organizations or CMOs (35%), and public-private partnerships (20%), where in-house dominates as majors like Takeda invest in owned facilities for supply security, CMOs offer flexibility for complex formulations, and partnerships aid rare disease and pandemic prep.
By technology platform, traditional batch manufacturing holds 35%, continuous manufacturing 25%, bioprocessing and single-use systems 20%, mRNA and advanced platforms 10%, and automation/AI-enabled 10%, shifting towards continuous and AI for efficiency amid PMDA compliance needs.
By therapeutic focus, oncology leads at 30%, followed by cardiovascular diseases (20%), infectious diseases (15%), rare diseases (15%), neurology (10%), and others (10%), driven by Japan's aging population and oncology R&D strength.
By facility type, large-scale production facilities claim 40%, mid-scale specialized 35%, and small-scale/high-potency 25%, catering to mass biologics, niche specialties, and personalized meds respectively.
By end-user, domestic pharma companies hold 50%, multinationals 25%, CDMOs 15%, and research/government 10%, prioritizing self-reliance.
Looking For A Detailed Full Report? Get it here:
https://www.datamintelligence.com/buy-now-page?report=japan-pharmaceutical-manufacturing-localization-market
Regional Insights
Japan's pharmaceutical manufacturing localization concentrates in the Kanto region, particularly around Tokyo and Yokohama, where over 50% of activity occurs due to clustered R&D centers, port access for exports, and headquarters of leaders like Takeda and Astellas, fostering innovation in biologics and APIs. Kansai, including Osaka and Kobe, follows with strong biotech hubs and CMO expansions, benefiting from historical pharma presence and government-backed regenerative medicine initiatives.
Hokkaido and Kyushu emerge as high-growth areas for specialized facilities, with Hokkaido leveraging cold-chain logistics for vaccines and Kyushu attracting investments in high-potency APIs via tax incentives and lower costs. Overall, urban hubs drive 80% of localization efforts, supported by METI subsidies, while rural expansions address capacity for cell/gene therapies.
Get Customization in the report as per your requirements:
https://www.datamintelligence.com/customize/japan-pharmaceutical-manufacturing-localization-market
Market Drivers
Japan's push for pharmaceutical manufacturing localization stems from lessons of global disruptions like COVID-19, prompting prioritization of domestic APIs and essentials to ensure uninterrupted supply for its aging population, which drives chronic disease demand. Government policies from the Ministry of Health, Labour and Welfare (MHLW) and METI offer tax benefits, subsidies, and streamlined PMDA approvals, accelerating onshoring of biologics and vaccines production. Technological shifts towards continuous manufacturing, AI quality control, and single-use systems boost efficiency, while rising oncology and rare disease needs fuel investments in advanced facilities.
Market Restraints
High capital costs for GMP-compliant facilities and cutting-edge bioprocessing tech pose barriers, especially for SMEs amid Japan's stringent regulations and land scarcity in key regions. Skilled labor shortages in advanced biomanufacturing, despite workforce programs, slow scaling, compounded by competition from lower-cost Asian CMOs. Regulatory hurdles for novel therapies like cell/gene products, though easing, still demand extensive validation, delaying localization timelines.
Market Opportunities
Expansion in high-growth segments like cell/gene therapies and mRNA platforms offers vast potential, backed by Japan's regenerative medicine leadership and post-pandemic vaccine infrastructure. Public-private partnerships and CDMO collaborations enable smaller players to localize without full capex, while export focus to APAC leverages Japan's quality reputation for specialty drugs. Digital twins, predictive analytics, and green manufacturing align with sustainability mandates, opening doors for efficiency gains and new funding.
Reasons to Buy the Report
✔ Comprehensive market sizing, forecasts to 2033, and CAGR analysis from DataM Intelligence.
✔ Detailed segmentation by product, model, tech, therapeutics, and end-user with share insights.
✔ Competitive landscape profiling top players like Takeda and Astellas with strategies.
✔ In-depth drivers, restraints, opportunities, and recent developments for strategic planning.
✔ Regional trends, policy impacts, and tech innovations for investment decisions.
Frequently Asked Questions (FAQs)
◆ How big is the Japan pharmaceutical manufacturing localization market?
◆ What is the projected growth rate of the Japan pharmaceutical manufacturing localization market?
◆ Who are the key players in the Japan pharmaceutical manufacturing localization market?
◆ What is the market forecast for the Japan pharmaceutical manufacturing localization market in 2033?
◆ Which region dominates the Japan pharmaceutical manufacturing localization market?
Company Insights
Key players operating in the Japan pharmaceutical manufacturing localization market include:
Takeda Pharmaceutical Company Limited
Astellas Pharma Inc.
Daiichi Sankyo Company, Limited
Otsuka Pharmaceutical Co., Ltd.
Eisai Co., Ltd.
Mitsubishi Chemical Holdings Corporation
Fujifilm Diosynth Biotechnologies
CMIC Group
Sysmex Corporation
FUJIFILM Corporation
Recent Developments
✅ In February 2026, Hisamitsu Pharmaceutical announced a management buyout valued at approximately $2.55 billion to enhance strategic flexibility in pain management therapeutics. The move is expected to accelerate domestic innovation and localized production of transdermal and neuropathic pain treatments. This reflects Japan’s push toward strengthening internal pharmaceutical capabilities.
✅ In January 2026, Samsung Biologics expanded its footprint in Japan by strengthening partnerships with local pharmaceutical firms. The company’s Tokyo presence aims to support biologics manufacturing and collaboration, contributing to localization of drug production including pain therapeutics. This aligns with Japan’s broader strategy to enhance domestic manufacturing capacity.
✅ In December 2025, Moderna continued operations of its mRNA research and manufacturing facility in Kanagawa despite canceling a larger plant project. The existing facility supports localized drug development and production capabilities in Japan, including potential applications in pain-related therapies. This reflects ongoing adjustments in Japan’s pharmaceutical manufacturing environment.
Enquire Now for Custom Research:
https://www.datamintelligence.com/custom-research
Conclusion
The Japan pharmaceutical manufacturing localization market stands at a pivotal juncture, propelled by resilience needs, policy support, and tech advancements to secure a US$21.33 billion future by 2033. Stakeholders investing in biologics, smart manufacturing, and regional hubs will capture growth in this vital sector, ensuring Japan's pharma self-sufficiency amid global uncertainties. For tailored insights, DataM Intelligence reports deliver actionable intelligence.
Sai Kiran
DataM Intelligence 4market Research LLP
+1 877-441-4866
sai.k@datamintelligence.com
Visit us on social media:
LinkedIn
YouTube
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.